Cellular Mechanisms Behind Diabetes-Derived Vascular Disease Unveiled
Diabetes affects over half a billion people globally. Along with direct consequences to those with the disease, it also contributes to and predisposes affected individuals to a host of other conditions. Specifically, it is a known contributing factor in the development of vascular disease, including peripheral artery disease. While therapies exist, they are not very effective, and peripheral artery disease can lead to restricted blood flow in peripheral limbs, which sometimes leads to amputation. Understanding the mechanism driving the connection at the tissue and cellular level has the potential to improve therapy options and the development of new treatments.
Normal function of the peripheral vasculature requires communication and cooperation between the vascular endothelium and macrophages. “Monocytes patrol the vascular endothelium and remove damaged cells, and intimal-resident macrophages maintain a nonthrombogenic endothelial state,” wrote the authors of a study led by Zhen Chen, PhD, at City of Hope. They explained that under stress, macrophages can modulate vascular remodeling and in certain conditions, like cancer, they “can secrete inflammatory mediators to disrupt endothelial cell tight junctions and increase endothelial cell permeability.”
The team decided to explore the cellular cross-talk between macrophages and endothelial cells, as well as the resulting vascular function, to better understand the mechanisms behind peripheral artery disease induced by diabetes.
They published their work in a paper titled “Diabetes-induced TREM2–endothelial cell signaling impairs ischemic vascular repair” in Science Translational Medicine.
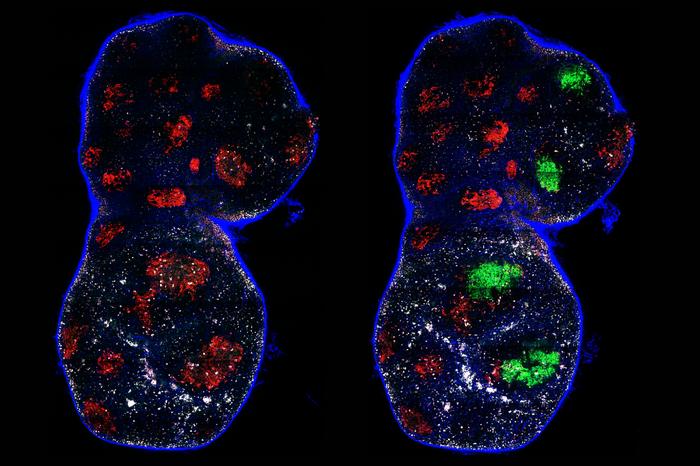
Using samples collected post-mortem from either donors with type 2 diabetes or donors without diabetes, the researchers aimed to systematically map the interactions between macrophages and endothelial cells in the arterial wall. They “leveraged single-cell RNA sequencing and spatial transcriptomics to profile human mesenteric arteries…generating a transcriptome and interactome atlas of diabetic vasculature.”
Their analysis identified increased expression in paired genes between macrophages and endothelial cells. Triggering receptor expressed on myeloid cells 2 (TREM2) is a gene previously identified as connected with metabolic disease, atherosclerosis, cancer, and neurodegeneration. In macrophages, TREM2 had increased expression in tissues from donors with type 2 diabetes, with concurrent expression of the TREM2 ligands in endothelial cells. Macrophages with increased TREM2 presented with a foamy cell structure, indicative of a pro-inflammation phenotype. Additionally, these cells had a proinflammatory gene profile.
Inhibiting TREM2 in vitro resulted in proinflammatory responses in macrophages and endothelial cells, along with increased migration of endothelial cells. To replicate TREM2 inhibition in vivo, the researchers used a mouse model for diabetes with hindlimb ischemia and treated them with a neutralizing antibody. This resulted in symptom improvement and improved blood vessel flow. Alternatively, activation of TREM2 with an agonist resulted in reduced blood flow and vascular damage.
Further analysis of human donor samples “confirmed elevated endothelial cell TREM2 signaling in human peripheral arterial disease, particularly in the setting of diabetes mellitus, highlighting its translational relevance.”
Together, these data show how TREM2 is involved with macrophage-endothelial cell communication within the peripheral vasculature. The authors pointed out that while TREM2 is a therapeutic target in treating Alzheimer’s disease and cancer, it might also have utility in treating peripheral artery disease.
“Plasma sTREM2 may be useful as a circulating marker of endothelial/vascular dysfunction in peripheral artery disease for risk stratification and outcome prediction,” they wrote. “In addition, our findings suggest caution when considering TREM2-enhancing therapeutics, particularly in individuals with existing diabetes mellitus and ischemic disease.”
This work underscores the need for more research into the details of disease mechanistic function to both better understand the cause of disease and to identify potential therapeutic targets.
“Future studies will need to dive deeper into how insulin deficiency or resistance and hyperglycemia activate macrophages to augment TREM2 expression and induce vascular dysfunction,” wrote Michael Chang, Michael T. Patterson, PhD, and Jesse Williams, PhD, in a related focus.
“Overall, the work of Malhi et al. advances our mechanistic understanding of type 2 diabetes-driven peripheral artery disease and has laid the foundation for developing targeted therapies for a disease with few viable treatment options,” they concluded.
The post Cellular Mechanisms Behind Diabetes-Derived Vascular Disease Unveiled appeared first on GEN - Genetic Engineering and Biotechnology News.
Apa Reaksi Anda?
 Suka
0
Suka
0
 Kurang Suka
0
Kurang Suka
0
 Setuju
0
Setuju
0
 Tidak Setuju
0
Tidak Setuju
0
 Bagus
0
Bagus
0
 Berguna
0
Berguna
0
 Hebat
0
Hebat
0