Novel KIR‑CAR T Approach Shows Early Activity Against Solid Tumors

CAR T cell therapies have revolutionized outcomes for certain blood cancers, yet their impact on solid tumors has lagged. The field has long wrestled with T cell exhaustion—a state in which engineered cells lose their potency and fail to sustain an anti‑tumor response.
At this year’s AACR annual meeting in San Diego, researchers from the Perelman School of Medicine at the University of Pennsylvania presented first‑in‑human Phase I data pointing to a possible solution. Their novel “KIR‑CAR” T cell therapy demonstrated a favorable safety profile and early signals of activity across multiple solid tumor types.
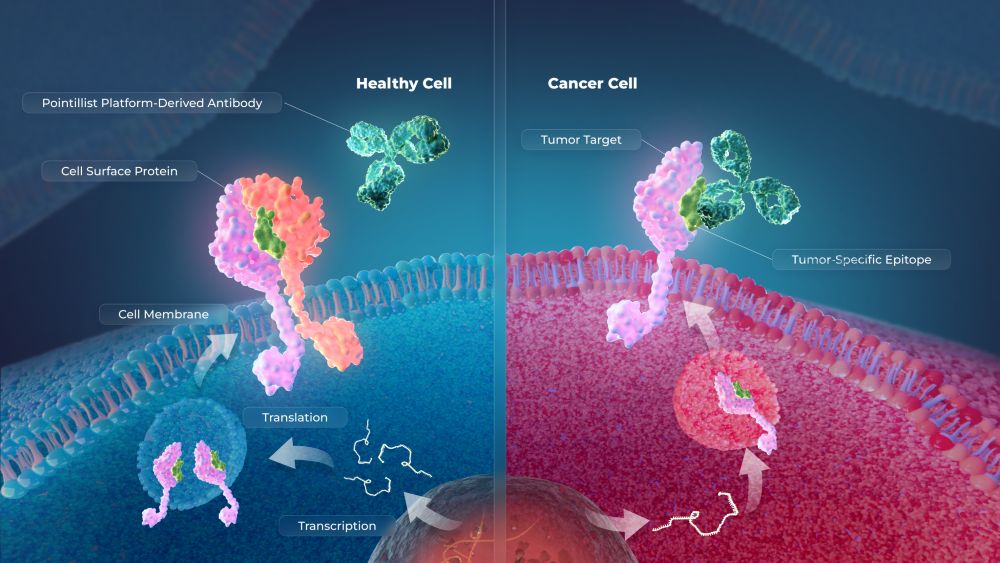
The investigational therapy, SynKIR-110, represents a departure from traditional CAR T designs. Rather than using a single-chain receptor, the therapy is modeled after natural killer (NK) cell receptors and uses a “multi-chain” architecture.
This design separates tumor recognition from activation, effectively creating an intrinsic “on-off” mechanism. The T cell remains in a resting state until it encounters its target, at which point the receptor components assemble to trigger an immune attack.
“The KIR-CAR design provides a natural ‘on-off’ mechanism, which helps avoid the problem of T cell exhaustion,” said Janos L. Tanyi, MD, PhD, principal investigator of the study. “The CAR turns on when it finds its target, kills it, and then rests, rather than constantly burning energy.”
This contrasts with conventional CAR T cells, which remain continuously active and can become depleted over time, limiting their effectiveness—particularly in the more complex microenvironment of solid tumors.
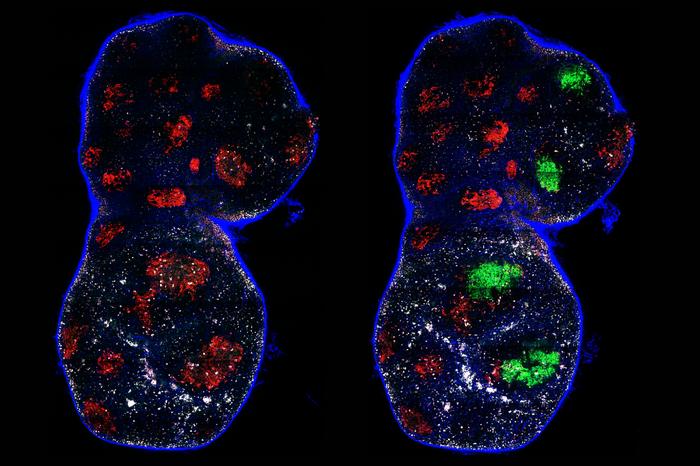
The Phase I dose-escalation trial enrolled nine patients with advanced, mesothelin-expressing cancers, including ovarian cancer, mesothelioma, and cholangiocarcinoma. These patients had limited treatment options, having received an average of four prior lines of therapy.
Although the primary goal of the study was to assess safety, early signs of efficacy were observed. Disease stabilization was reported in four patients, and one patient in the highest dose cohort achieved an ongoing partial response.
“These are cancer types that have never had an approved cell therapy,” Tanyi said. “We’re seeing good efficacy signals, even at low doses, and limited toxicity.”
The results suggest that the therapy may be able to generate meaningful anti-tumor responses even in heavily pretreated populations.
Safety has been another major barrier for CAR T therapies, particularly in solid tumors. However, the KIR-CAR approach appears to mitigate some of these concerns.
No dose-limiting toxicities were observed in the initial cohorts. Cytokine release syndrome (CRS), a common side effect of CAR T therapy, occurred in 33% of patients but was limited to low-grade events. Notably, there were no cases of immune effector cell-associated neurotoxicity syndrome (ICANS), a more severe complication sometimes seen with CAR T therapies.
The ability to limit toxicity while maintaining activity is a key step toward broader application of cell therapies in solid tumors.
SynKIR-110 targets mesothelin, a protein expressed on the surface of several solid tumors but largely absent from normal tissues. This makes it an attractive target for immunotherapy, particularly in cancers such as ovarian cancer and mesothelioma, where treatment options are limited.
The trial results indicate that the therapy’s activity is not confined to a single tumor type, raising the possibility of broader applicability across mesothelin-expressing cancers.
The findings come amid growing efforts to adapt CAR T technology for solid tumors. While the approach has revolutionized hematologic malignancies, solid tumors present additional challenges, including immunosuppressive microenvironments, physical barriers to T cell infiltration, and antigen heterogeneity.
Researchers are exploring multiple strategies to address these barriers, including improved targeting, combination therapies, and next-generation receptor designs such as KIR-CAR.
As noted by CAR T pioneer Carl June, MD, advancing cellular therapies into solid tumors remains a central goal for the field.
The Phase I study continues to enroll patients, aiming for a 42‑person cohort to define the maximum tolerated dose ahead of Phase II. Early readouts show that CAR T expansion rises with dose, a pattern that may translate into stronger anti‑tumor activity at higher levels.
While still preliminary, the findings highlight the potential of multi‑chain CAR designs to sustain activity without added toxicity. If confirmed, KIR‑CAR therapies could usher in a new generation of engineered immune cells that more closely mirror natural immune regulation.
For now, the data offer a promising sign that CAR T innovation may finally be gaining ground in solid tumors.
The post Novel KIR‑CAR T Approach Shows Early Activity Against Solid Tumors appeared first on GEN - Genetic Engineering and Biotechnology News.
Apa Reaksi Anda?
 Suka
0
Suka
0
 Kurang Suka
0
Kurang Suka
0
 Setuju
0
Setuju
0
 Tidak Setuju
0
Tidak Setuju
0
 Bagus
0
Bagus
0
 Berguna
0
Berguna
0
 Hebat
0
Hebat
0