Breaking Through the Barrier

According to the American Brain Foundation, over one in three people around the world are affected by neurological conditions, the leading cause of illness and disability worldwide. This silent epidemic is not country-specific. Neurological conditions such as lysosomal storage disorders, rare enzyme deficiencies, and Alzheimer’s and Parkinson’s disease take their victims, regardless of age, race, or location.
For decades, scientists have struggled to deliver therapeutics to the brain, only to be thwarted by the highly protective blood-brain barrier (BBB). First-generation approaches demonstrated proof of principle but still require advancements to improve the ability to reach specific areas of the brain, or specific cell types, safely, and with sufficient dosage to enable meaningful therapeutic effects.
Although much remains unknown generally about brain biology and its defensive mechanisms, novel therapies for devastating neurological diseases are progressing into clinical trials. There is no magic bullet—no promises, no cures—but a gleaming light can be seen in this particular long and dark tunnel.
Dedicated scientists continue to work on gene therapies for the indications that most benefit from a once-and-done approach, in addition to neurological shuttles to address those disorders that require therapeutic tempering and dosage control.
Expanding platform technologies
In 2021, JCR Pharmaceuticals received regulatory approval for the first biotherapeutic, IZCARGO (pabinafusp alfa), designed to cross the BBB to deliver a therapeutic enzyme for the treatment of a lysosomal storage disorder called mucopolysaccharidosis type II (MPS II) or Hunter syndrome.
(pabinafusp alfa), designed to cross the BBB to deliver a therapeutic enzyme for the treatment of a lysosomal storage disorder called mucopolysaccharidosis type II (MPS II) or Hunter syndrome.
The platform technology has been expanded to exploit receptor-mediated transcytosis (RMT) to address other lysosomal storage and neurodegenerative diseases. Still, delivery to specific cells or parts of the brain remains challenging, along with efficient delivery of antisense oligonucleotides or siRNA.
“The issue is not delivery across the BBB, but the endosomal escape to efficiently suppress the target RNA,” said Hiroyuki Sonoda, PhD, representative director, president, and CSO, at JCR Pharmaceuticals. “Small molecule CNS delivery is related to physicochemical properties. The structural design needs to make them lipophilic, yet also able to evade typical transporter clearing mechanisms.”
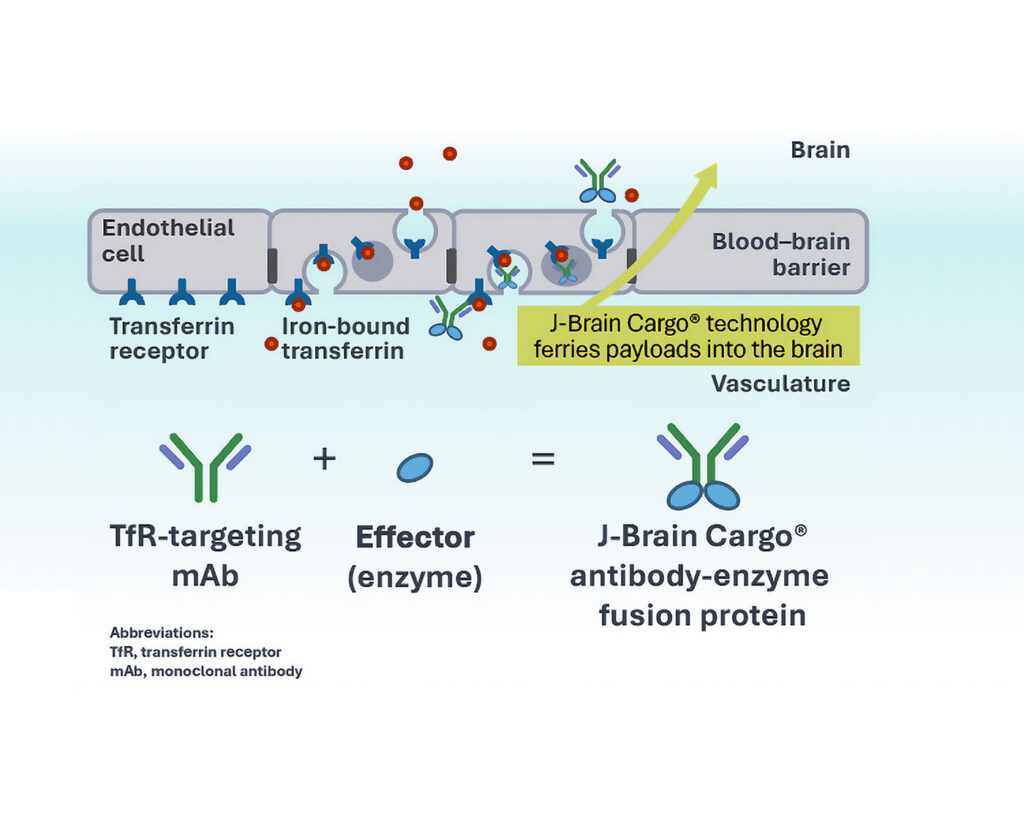
J‑Brain Cargo® uses RMT, mainly focusing on the transferrin receptor (TfR). Other promising candidates target different receptors. “We have successfully transported enzymes, antibodies, peptides, decoy receptors, antisense oligos, and siRNA into the CNS,” commented Sonoda. J‑Brain Cargo is particularly suited for enzyme replacement therapies in lysosomal storage disorders and conditions where dose control, reversibility, and titration are important.
For gene therapies, JCR developed the JUST-AAV platform technology. Novel changes in the capsid almost completely eliminate liver tropism. The modified capsids express miniaturized antibodies on the capsid surface against receptors on selected tissues, organs, or the BBB, enhancing targeted delivery. JUST‑AAV is for diseases where continuous transgene expression is desired to achieve the optimal effect.
Several candidates are in global clinical trials, including JR-141 (pabinafusp alfa) for individuals with MPS II (also known as Hunter syndrome), JR-171 to treat MPS I (also known as Hurler, Hurler Scheie, or Scheie syndromes), and JR-441 for individuals with MPS IIIA (also known as Sanfilippo syndrome A).
Programs in collaboration with MEDIPAL HOLDINGS CORPORATION are in different stages of clinical and pre-clinical development for individuals with MPS IIIB (also known as Sanfilippo syndrome B), Fucosidosis, and GM2 gangliosidosis (including Tay-Sachs and Sandhoff disease).
Collaborating with leading pharmaceutical companies is core to JCR’s strategy to bring these platform technologies to broader application. “We enable our partner by turning their biologics into CNS-penetrating versions of their original molecule,” said Sonoda.
JCR manufactures most of its drug products in-house. Last year, they were selected for the Ministry of Economy, Trade and Industry’s “Regenerative CDMO Subsidy” to expand biomanufacturing capacity for regenerative, cell, and gene therapies.
Optimizing BBB transport
“Protein engineering architecture differentiates our delivery technology along with its optimization for efficacy, safety, and tolerability,” said Ryan Watts, PhD, co-founder and CEO of Denali Therapeutics.
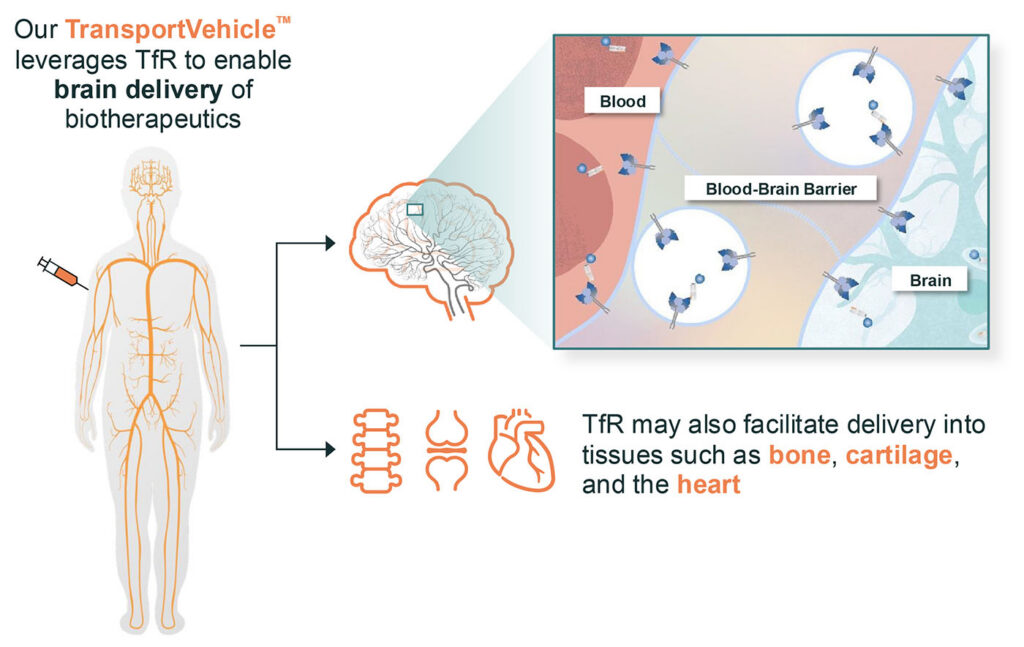
The TransportVehicle (TV) technology has the RMT binding site integrated directly into the constant domain (Fc) of an antibody for optimal properties and modularity. This allows the same TV sequences to transport a range of large molecule biotherapeutics such as enzymes, oligonucleotides, and antibodies for systemic administration. The engineered Fc domains bind to specific natural transport receptors expressed at the BBB, such as TfR.
(TV) technology has the RMT binding site integrated directly into the constant domain (Fc) of an antibody for optimal properties and modularity. This allows the same TV sequences to transport a range of large molecule biotherapeutics such as enzymes, oligonucleotides, and antibodies for systemic administration. The engineered Fc domains bind to specific natural transport receptors expressed at the BBB, such as TfR.
“Our research recently demonstrated that a TV platform-enabled anti-Ab antibody improved distribution in the brain and significantly reduced risk of Amyloid-Related Imaging Abnormalities (ARIA) in a mouse model of Alzheimer’s disease, when compared with a conventional anti-Ab antibody.1 The study provides the first mechanistic insight for mitigating the risk of ARIA,” detailed Watts.
The Enzyme TransportVehicle (ETV) contains a fusion of a therapeutic enzyme. The Fc portion of the fusion molecule binds the apical surface of the TfR to avoid interference with normal iron transport.
In March 2026, Denali’s lead ETV program, Avlayah (tividenofusp alfa-eknm), received FDA accelerated approval for the pediatric treatment of the lysosomal storage disorder MPS II. Avlayah is the foundation for their broader ETV franchise, addressing other lysosomal storage disorders such as MPS IIIA. Results from the open-label Phase I/II clinical trial are available.2
(tividenofusp alfa-eknm), received FDA accelerated approval for the pediatric treatment of the lysosomal storage disorder MPS II. Avlayah is the foundation for their broader ETV franchise, addressing other lysosomal storage disorders such as MPS IIIA. Results from the open-label Phase I/II clinical trial are available.2
Their Oligonucleotide TransportVehicle (OTV) platform is an engineered TV conjugated to an oligonucleotide for the systemic delivery of genetic medicines to the brain. Extensive characterization and research demonstrate the ability of OTV to elicit broad biodistribution of oligonucleotide therapies throughout the CNS following systemic exposure.
“For example, our investigational therapy DNL628 for the treatment of Alzheimer’s disease is designed to cross the BBB and reduce the tau protein by targeting the MAPT gene that encodes for tau,” explained Watts.
Lastly, the Antibody TransportVehicle (ATV) platform is designed to enable brain delivery of antibodies capable of selective immune activation and a targeted therapeutic approach after intravenous administration. The investigational anti-Ab antibody therapy DNL921, for example, is designed to reduce amyloid plaques and avoid ARIA.
The TV-enabled clinical development portfolio also includes candidates for frontotemporal dementia-granulin and Pompe disease.
Advancing clinical options
“It is exciting to begin to see that delivery through the BBB is possible using gene therapy or shuttle approaches,” said Todd Carter, PhD, CSO at Voyager Therapeutics. Although first-generation therapeutics are demonstrating meaningful levels of delivery, optimization, and improvement of the functionality, exposure duration, and therapeutic effects are still needed.
“For some diseases, gene therapy is the preferred treatment modality, as both the capsid and the payload can be modified to perform a specific job,” said Carter. But viral vector delivery for gene therapy has had problems with liver-based toxicity.
For the best human translation opportunities, Voyager developed a model in non-human primates (NHPs) requiring cross-species activity across multiple NHP species. This strategy resulted in the company’s TRACER (Tropism Redirection of AAV by Cell-type-specific Expression of RNA) technology, used to screen tens of millions of vector variants using barcoded libraries in which capsids were modified with slight insertions of seven to nine amino acids.
(Tropism Redirection of AAV by Cell-type-specific Expression of RNA) technology, used to screen tens of millions of vector variants using barcoded libraries in which capsids were modified with slight insertions of seven to nine amino acids.
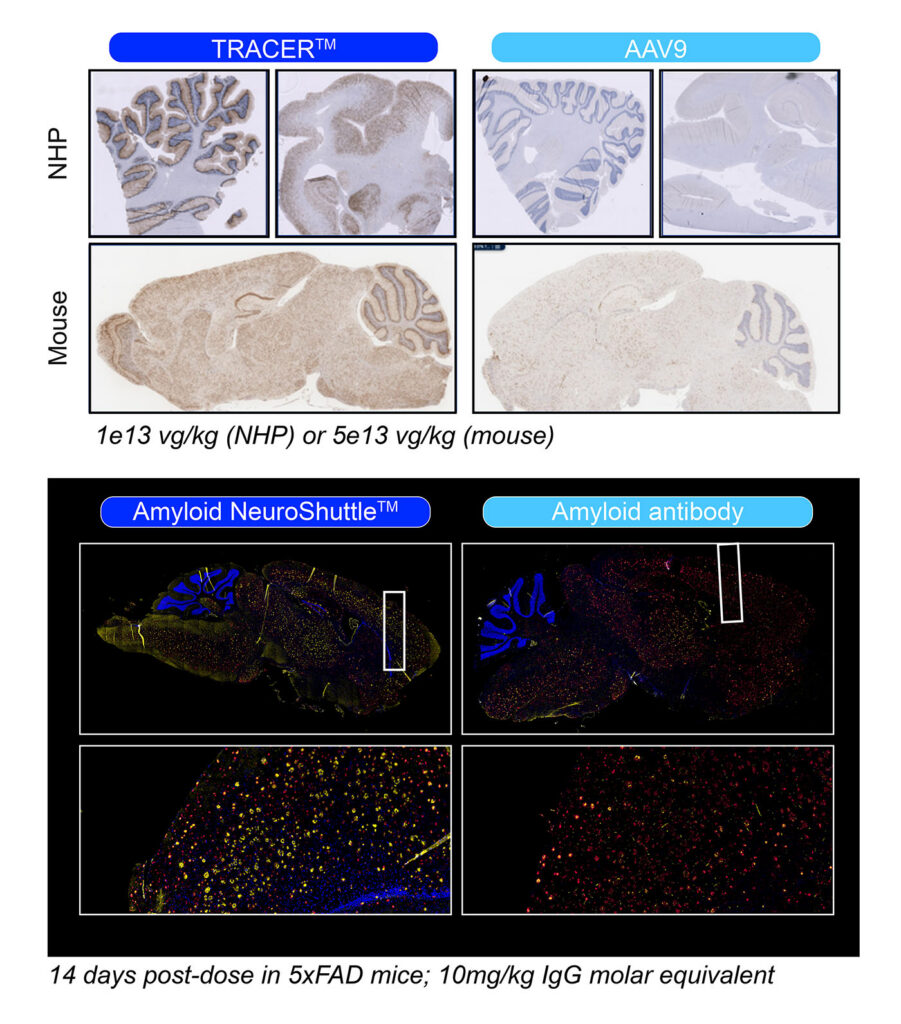
Successful expression in neurons demonstrated that the capsids crossing the BBB worked. Directed evolution improved them. “Next, we needed to determine the mechanism—the receptors they were targeting,” said Carter. This led to the identification of the receptor, alkaline phosphatase (ALPL), tissue nonspecific.
Now, Voyager has multiple families of capsids that mediate delivery into the brain, are detargeted from the liver, and, for the most advanced, have improved the capsid’s ability to target the brain using ALPL. “Using the ALPL receptor elevates delivery to the brain and allows us to substantially reduce dosage,” said Carter.
“I would not have picked ALPL just on face value,” added Mihalis Kariolis, PhD, vice president of non-viral therapeutics at Voyager Therapeutics. “It highlights the power of the unbiased TRACER approach. Expanding the number of brain delivery receptors provides highly differentiated options to reduce side effects and expand the diversity of treatment modalities.”
Both gene therapy and shuttle approaches have opportunities in different indications. Once-and-done gene therapy is not tweakable, whereas shuttle-based dosing is. “In our APOE gene therapy program, we want to reduce existing APOE4 and replace it with APOE2 permanently,” said Carter. “The shuttle has advantages in situations where permanent ongoing delivery is not required.”
Voyager’s most advanced program (VY7523) is a tau monoclonal antibody that is exquisitely specific for pathological tau. Data will be available in the second half of the year. A gene therapy (VY1706) moving into the clinic this year is designed to knock down tau mRNA and protein intracellularly. A collaboration with Neurocrine Biosciences focuses on Friedreich’s ataxia (FA) and is also expected to enter the clinic this year.
Combining transport receptors
The protective BBB is crucial for maintaining homeostasis and ensuring proper neurological function. Comprised of both cellular and acellular components, this sophisticated structure tightly regulates information flow between the periphery and the brain. According to Tanya Wallace, PhD, vice president of neuroscience discovery research at AbbVie, despite the BBB’s importance, many seemingly basic biological questions remain unanswered, fueling additional global research.
The complexity of the BBB also represents a significant bottleneck for advancing therapeutics targeting brain-related disorders. Historically, achieving therapeutically relevant levels of drugs in the brain has been a major challenge in treating serious diseases such as Alzheimer’s and Parkinson’s diseases. “A notable success story is the development of L-DOPA, a prodrug that leverages existing transport mechanisms to cross the BBB,” said Wallace. Once in the brain, L-DOPA is metabolized into dopamine, offering a key symptomatic treatment for Parkinson’s disease.
Breakthroughs in delivery now allow scientists to leverage more technologies that can bring not only small molecules but also complex biologics into the brain. The Modular Delivery (MODELTM) platform exemplifies this progress. The platform enables engineering of bispecific antibodies, capable of targeting naturally expressed BBB receptors such as TfR and CD98. TfR and CD98 are well-characterized at the BBB, and, together, they offer distinct advantages for increasing brain exposure to therapeutics.
“By engaging these transport pathways, the platform can enhance the uptake of a variety of therapeutics, including antibodies and oligonucleotides,” highlighted Wallace. “This multi-receptor strategy provides flexibility to optimize the balance of uptake, release, and distribution in the brain, paving the way for potentially more effective treatments across neurological disease areas.”
This platform technology facilitated the development of ABBV-1758, which is progressing in clinical development. ABBV-1758 utilizes TfR to transport a 3pE-Ab antibody across the BBB to enable the removal of amyloid beta plaques, a pathological hallmark of Alzheimer’s disease.
As scientists aspire to further refine delivery strategies, ongoing research is exploring additional receptors and innovative approaches, including insulin-like growth factor 1 receptor (IGF-1R) and brain cell-type-specific targeting. The field is rapidly evolving to advance more precise, personalized interventions for challenging neuroscience conditions.
“Successful brain delivery requires more than just advances in transport technology; it demands interdisciplinary collaboration, novel preclinical models, and thoughtful clinical translation,” Wallace pointed out. Continued biological research and investment into innovative discovery platforms will be crucial for bringing transformative therapies to patients with the greatest unmet needs.
References
- Pizzo ME, Plowey ED, Khoury N et al. Transferrin receptor-targeted anti-amyloid antibody enhances brain delivery and mitigates ARIA. Science. 2025 Aug 7;389(6760):eads3204. doi: 10.1126/science.ads3204.
- Muenzer J, Burton BK, Harmatz P et al. An intravenous brain-penetrant enzyme therapy for mucopolysaccharidosis II. N Engl J Med. 2026 Jan 1;394(1):39-50. doi: 10.1056/NEJMoa2508681.
The post Breaking Through the Barrier appeared first on GEN - Genetic Engineering and Biotechnology News.
Apa Reaksi Anda?
 Suka
0
Suka
0
 Kurang Suka
0
Kurang Suka
0
 Setuju
0
Setuju
0
 Tidak Setuju
0
Tidak Setuju
0
 Bagus
0
Bagus
0
 Berguna
0
Berguna
0
 Hebat
0
Hebat
0