Going Non-Viral: Gene Delivery Enters Its Translation Era

CEO, BreezeBio
Kunwoo Ryan Lee, PhD, knew as early as 2012 that solving the delivery problem would be crucial in fulfilling the promise of the newly discovered CRISPR-Cas9 gene editing technology. He felt strongly that gene editing had potential to transform medicine by curing genetic disorders, but the viral and non-viral vectors available at the time had significant drawbacks. With the support of CRISPR pioneers Jennifer Doudna and Stanley Qi, Lee completed his doctoral thesis on a gold nanoparticle delivery system for Cas9 ribonucleoprotein. He went on to co-found BreezeBio, formerly GenEdit, in 2016 with the aim of creating the next generation of gene editing-based therapeutics. To realize that goal, Lee and his team looked beyond traditional viral gene delivery systems and instead invented a new technology from the ground up.
Most clinical gene therapy trials use viral vectors, including retroviruses, lentiviruses, adenoviruses, and adeno-associated viruses. However, viral vectors are limited in the size of the gene they can deliver. They also tend to trigger strong immune reactions and usually can’t be dosed more than once due to acquired immunity.
Non-viral vectors are an alternative technology that offer greater gene loading capacity, more straightforward preparation, and less likelihood of triggering problematic immune reactions. BreezeBio and other biotechnology companies are reimagining gene delivery through non-viral approaches like targeted LNPs, transposons, and novel chemistry.

“Using the platform, we have demonstrated that we can deliver to the spleen, immune system, heart, and lung,” Lee said. The firm also developed a set of nanoparticles targeted to the central nervous system.
Based on those targeted delivery profiles, the Brisbane, California-based BreezeBio has worked with multiple partners to provide delivery solutions for their products, including a multiyear collaboration with Genentech, a member of the Roche Group, signed in 2024. Meanwhile, the company is also advancing its own pipeline of therapeutics built on the NanoGalaxy platform, including a lead candidate for type 1 diabetes, as well as investigational therapies for autoimmune disease and cancer.
Lee said a key advantage of the NanoGalaxy platform for their pipeline, which heavily leans toward autoimmune disease, is that, unlike a viral vector, the company’s studies have shown it does not activate the innate immune system. “That enabled us to use our technology for autoimmune applications and in more targeted oncology applications, as well,” Lee said.
Snug as a bug in a rug
The red flour beetle, a notorious scourge of grain and cereal stores, is the surprising source of Bio-Techne’s transposon-based, non-viral gene delivery system. The system, dubbed TcBuster for the beetle’s scientific name, Tribolium castaneum, was invented by B-MoGen, a spin-out of the University of Minnesota, which was acquired in 2019 by Minneapolis-based Bio-Techne. Researchers at B-MoGen and Bio-Techne developed a hyperactive version of the natural TcBuster transposon by creating a library of three million unique genetic variants and screening each in mammalian cells. In a proof-of-concept study, CAR NK cells engineered using TcBuster demonstrated in vitro functionality and improved survival in a preclinical model of Burkitt lymphoma with a single dose.1
“The reason you want a hyperactive version is that wild-type transposon systems are fairly low activity,” said Miles Smith, PhD, a product manager for cell and gene therapy at Bio-Techne. “For generating a cell therapy, you want something that’s going to be comparable to the state of the field, and that’s lentivirus.”
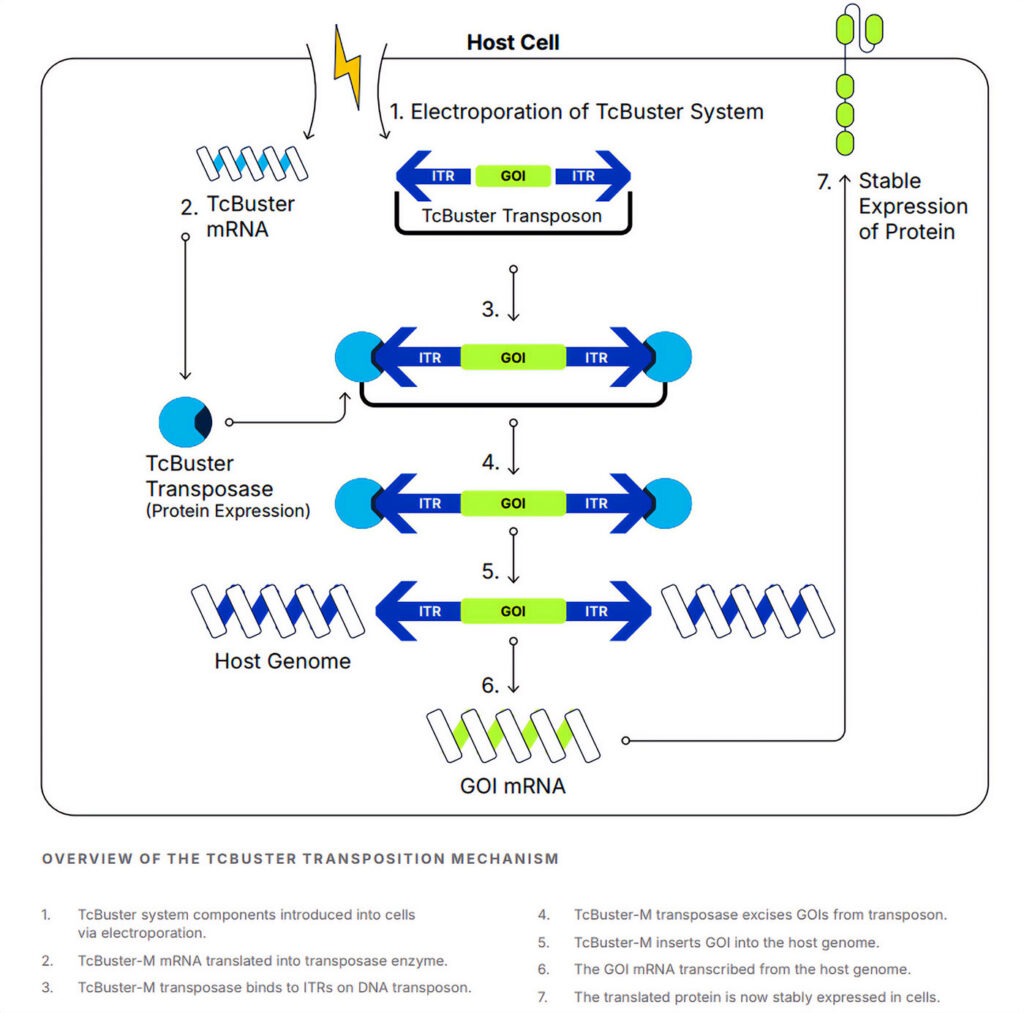
Unlike other commercial transposon systems for gene delivery, like Sleeping Beauty and PiggyBac, Smith said TcBuster is not restricted by exclusive licensing. “The turnaround time for GMP material is just a couple of months,” Smith said. “Versus something that might take a lot longer if you have to go through licensing or create a viral batch.”
Gene therapy SORTed
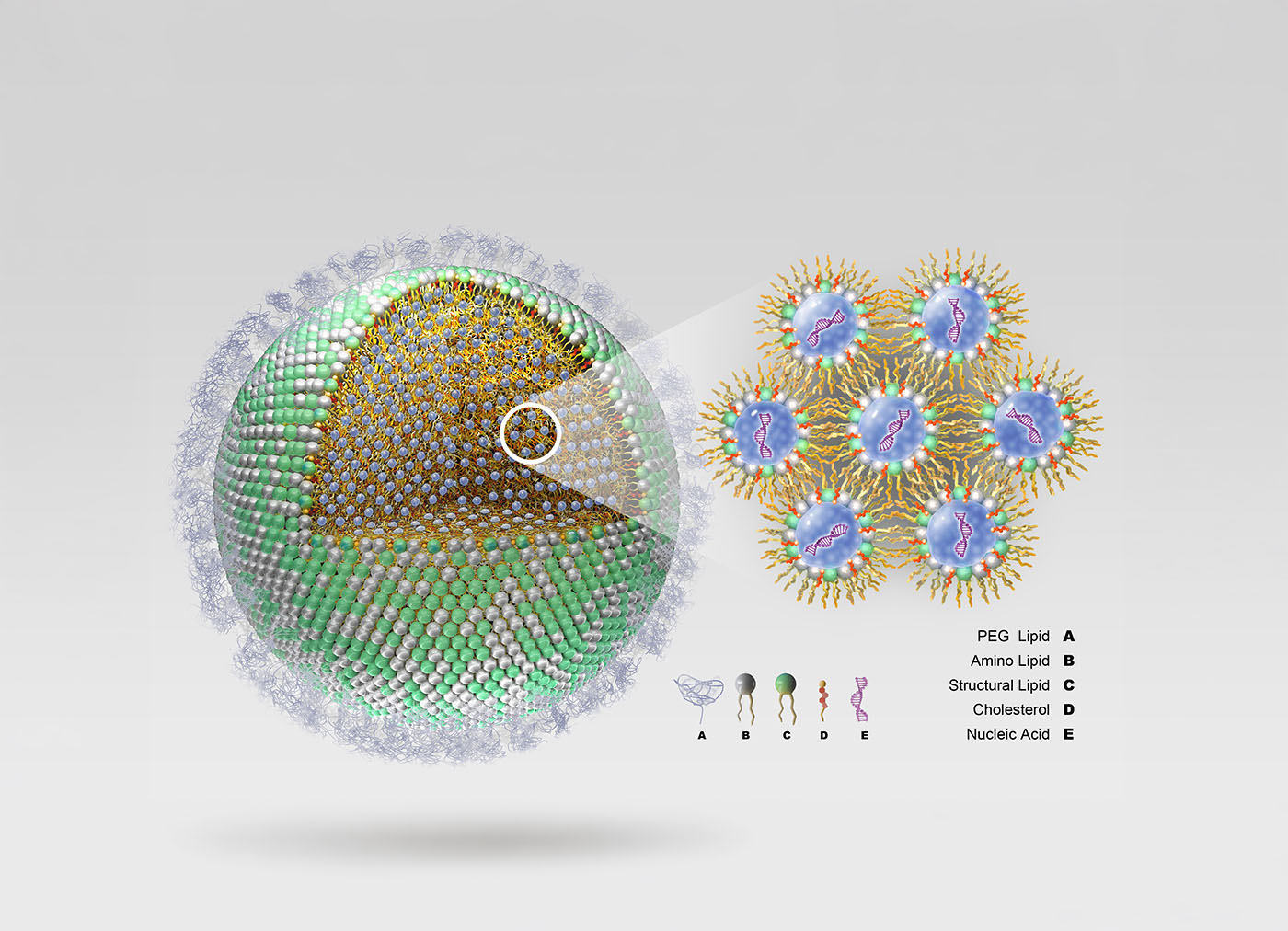
ReCode Therapeutics is developing a pipeline of genetic medicines based on its selective organ targeting (SORT) LNP platform, which adds an additional lipid to the standard LNP formulation, allowing it to zero in on specific organs. Conventional LNPs comprise four lipids—cholesterol, a helper phospholipid, a PEGylated lipid, and an ionizable lipid—that encapsulate a therapeutic gene cassette. These traditional LNPs are primarily taken up by the liver after intravenous administration, limiting their usefulness for other organs and systems. ReCode engineered its SORT LNPs with a biochemically distinct fifth lipid that enables the body to direct the particle to the targeted organ, such as the lung or spleen, bypassing the liver, if necessary.
“Because mRNA in a cell has a relatively short half-life, maybe a day or so, in order to have constant protein production, you need to administer it relatively frequently,” Vladimir Kharitonov, PhD, senior vice president of CMC and pharmaceutical sciences at ReCode, said. “With viral delivery, you can’t really administer it repeatedly.”

In 2024, ReCode presented preclinical data from its cystic fibrosis program showing its mRNA-based therapeutic RCT2100 significantly restored CFTR function in human bronchial epithelial cells derived from patients with cystic fibrosis. In vivo studies using a ferret model demonstrated improvement in mucociliary clearance. ReCode launched the first clinical trial of RCT2100 later that year. The LNP for RCT2100 contains SORT lipids to fine-tune its delivery to the airway epithelial cell types that have a defective or mutated CFTR protein, causing cystic fibrosis. The company is also developing a second mRNA therapy delivered via SORT LNP, RCT1100, for primary ciliary dyskinesia, which targets different cell types in the lung epithelium.
From idea to therapy faster
Gene delivery is just one of many services offered by GenScript to support research from discovery through clinical testing, including gene synthesis, CRISPR reagents, antibodies, and more.

Senior Director, GMP Manufacturing
GenScript
“Gene editing is entering a new era, and the focus has shifted from discovery to translation,” said Jianpeng Wang, PhD, senior director of nucleic acid and peptide R&D at GenScript. “Our goal at GenScript is to help scientists move from idea to therapy faster.”
When it comes to non-viral vectors, the firm offers off-the-shelf and bespoke solutions to fit the customer’s need for delivery of DNA, RNA, siRNA, peptides, and other molecules. Through its targeted LNP service, GenScript offers LNPs designed to enhance precision in directing genetic material to cells. GenScript’s ReadyEdit LNP solutions include Cas9 knock-in and knock-out, Cas12 knock-out, and base or prime editing tailored for the customer’s needs.
“In our ecosystem, we include all of the materials needed,” Wang said. “This integration can help scientists evaluate gene editing efficiency early, both ex vivo and in vivo.”
Wang said the choice of a vector is heavily dependent on the specific therapeutic program. “There isn’t a universally effective or better way to deliver a therapy, either viral or non-viral,” Wang said. He noted, for example, that viral vectors remain a good choice when long-term gene expression is desired. And for viral vectors, the manufacturing process might be more mature, easing transfer to a contract development and manufacturing organization.
However, Wang cautioned that viral vectors still present certain safety concerns. “In recent years, an increasing number of scientists and the FDA have recognized these risks,” he said, “leading to a surge in interest for non-viral delivery methods—particularly for in vivo CAR T therapy and gene editing.”
GenScript has provided LNP services to several customers globally. The most advanced of those is using GenScript’s GMP CRISPR materials (gRNA, HDR templates, and nuclease) alongside a customized LNP encapsulation recipe and is preparing an investigational new drug application.
Foundational LNP science
Vancouver-based Genevant traces its scientific lineage through a string of predecessor companies dating back to the early 2000s and controls foundational intellectual property for the field. Based on its scientists’ work at Protiva Biotherapeutics, the intellectual property comes to Genevant via Arbutus Biopharma, which acquired Protiva in 2015 and partnered with Roivant in 2018 to establish Genevant.
Unlike many companies developing nucleic acid delivery platforms that focus on a single payload modality, Genevant has applied its LNP to many payloads, including mRNA, siRNA, and gene editors in fields spanning antiviral, oncology, and metabolic disorders. The firm’s LNP platform is part of the first RNA-LNP product to achieve regulatory approval, Alnylam Pharmaceuticals’ Onpattro (patisiran), a treatment for polyneuropathy in people with hereditary transthyretin-mediated amyloidosis. Genevant’s LNP technology is also behind Moderna’s COVID-19 vaccines, which were confirmed earlier this month with the resolution of a longstanding patent dispute. An infringement case against Pfizer and BioNTech is pending. Genevant collaborated with Chula Vaccine Research Center and the University of Pennsylvania to develop a COVID vaccine for low- and middle-income countries in Southeast Asia during the pandemic. The program had success, demonstrating non-inferiority to Pfizer and BioNTech’s Comirnaty in clinical trials.
Some key differentiators for Genevant’s LNPs include strategies for optimized delivery in non-human primates instead of mice,2 which has resulted in improved gene editing in the liver, and novel chemistries for biodegradable LNPs that prevent accumulation in tissue.3 The company has recently disclosed data showing targeted delivery to T cells for in vivo CAR T therapy, as well as hematopoietic stem and progenitor cells (HSPC) and hepatic stellate cells.
References
- Skeate JG, Pomeroy EJ, Slipek NJ, et al. Evolution of the clinical-stage hyperactive TcBuster transposase as a platform for robust non-viral production of adoptive cellular therapies. Mol Ther. 2024;32(6):1817-1834. doi:10.1016/j.ymthe.2024.04.024
- Lam K, Schreiner P, Leung A, et al. Optimizing lipid nanoparticles for delivery in primates. Adv Mater. Published online March 27, 2023. doi: 10.1002/adma.202211420
- Holland, R, Lam K, Jeng S, et al. 2024. Silicon ether ionizable lipids enable potent MRNA lipid nanoparticles with rapid tissue clearance.” ACS Nano. 2024;18 (15): 10374–87. doi:10.1021/acsnano.3c09028.
The post Going Non-Viral: Gene Delivery Enters Its Translation Era appeared first on GEN - Genetic Engineering and Biotechnology News.
Apa Reaksi Anda?
 Suka
0
Suka
0
 Kurang Suka
0
Kurang Suka
0
 Setuju
0
Setuju
0
 Tidak Setuju
0
Tidak Setuju
0
 Bagus
0
Bagus
0
 Berguna
0
Berguna
0
 Hebat
0
Hebat
0