Method Identifies Cellular Makeup of Microenvironments Favoring Tumor Metastasis

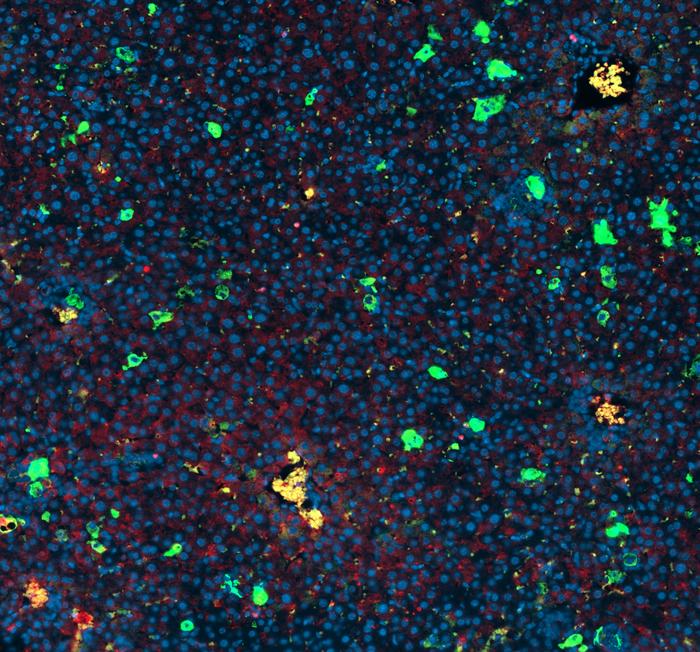
Researchers at Baylor College of Medicine and collaborating institutions have developed a method that reveals the cellular makeup of tissues that support metastatic cancer growth, which is the primary cause of death for most patients with solid tumors. The technique, sortase A-based microenvironment niche tagging (SAMENT), is designed to selectively label cells encountered by cancer cells during metastasis. The team’s tests using SAMENT not only revealed cellular features shared by metastatic niches of multiple cancer models but also uncovered an unexpected driver of immune suppression in bone metastasis.
“As tumors progress, cancer cells leave the original site and spread or metastasize to other organs where they seed new tumors,” said Xiang Zhang, PhD, William T. Butler, MD, Endowed Chair for Distinguished Faculty, professor of molecular and cellular biology, and director of the Lester and Sue Smith Breast Center at Baylor. “Our lab is interested in better understanding what cellular and molecular features support metastasis as these could guide the development of therapies to prevent, slow down, or eliminate them. In the current study, we first developed a new method to identify the makeup of metastatic niches.”
Zhang, also a member of Baylor’s Dan L Duncan Comprehensive Cancer Center, is senior and corresponding author of the team’s published paper in Cell, titled “Unbiased niche labeling maps immune-excluded niche in bone metastasis.”
During metastasis, cancer cells interact constantly with other normal cells in the body, and these interactions affect cell behavior, fate, and even response to therapies. “Numerous previous studies have elucidated the roles of specific microenvironment niches (i.e., cells that are immediately adjacent to cancer cells) in the progression of metastasis,” the authors wrote.
For their newly reported study the team developed the SAMENT technology. “Our method allowed us to identify specific cells encountered by cancer cells during metastasis,” said co-first author Fengshuo Liu, graduate student in the Cancer and Cell Biology Program working in the Zhang lab. “The method, called Sortase A–Based Microenvironment Niche Tagging (SAMENT), selectively labels normal cells that come into direct contact with cancer cells.”
The authors further explained, “By combining SrtA and synthetic ligand-receptor binding, we aim to label any cells that are physically encountered by cancer cells.”
The investigators’ tests using SAMENT revealed that pro-metastatic microenvironments of multiple cancer models in all the organs studied, including bone, lung, liver, and brain, shared common features, including an abundance of macrophage immune cells and shortage or absence of immune T cells, which typically help fight tumors. “Among all cell types, macrophages occur most frequently surrounding disseminated cancer cells and appear to be phenotypically re-programmed upon interaction with metastases,” they wrote.
Liu added, “However, bone metastases stood out. We were surprised to find that macrophages surrounding cancer cells in bone metastases activated a protein called estrogen receptor alpha (ERα). This protein is best known for its role in hormone-responsive breast cancer but is much less studied in macrophages or other immune cells.” The team added, “It also plays an important role in many other cell types, including macrophages, T cells, osteoblasts, and osteoclasts.”
The study showed that macrophages with active ERα signaling were not detected in normal bone or in primary tumors in other tissues. ERα-active macrophages were also present in human bone metastasis samples from patients with breast, lung and kidney cancers—including male patients. This showed that this finding is not limited to one cancer type or to women.
The researchers also investigated how cancer cells turned macrophages, which would typically fight cancer, into their allies. Cancer cells deliver small molecules called fatty acids (FAs) to macrophages, likely through tiny particles known as extracellular vesicles (EVs). These fatty acids activate a metabolic pathway in macrophages that turns on ERα signaling. “Taken together, our data indicate that ERα expression in macrophages is driven by cancer cell-derived FAs through paracrine interaction mediated by EVs,” they wrote.
Once ERα is active, macrophages become immunosuppressive—instead of helping the immune system attack cancer, they form a barrier that physically and chemically blocks T cells from reaching tumor cells. ERα-active macrophages act as bodyguards for metastatic cancer in bone.
“To test whether ERα in macrophages can drive bone metastasis, we genetically removed the ERα gene specifically from macrophages in mice,” Liu continued. “As a result, cancer cells were far less able to colonize bone in multiple cancer models. Tumors grew more slowly, and metastases in other organs that often arise from bone tumors were also reduced. Importantly, removing ERα from macrophages did not disrupt normal bone health—bone structure and remodeling remained intact.” In their paper the scientists stated, “Taken together, our results strongly support the hypothesis that ERα in macrophages plays an important role in bone colonization.”
“When macrophage ERα was genetically removed or when mice were treated with fulvestrant, an FDA-approved cancer drug that degrades estrogen receptors, T cells were able to enter metastatic lesions in bone and kill tumor cells,” Zhang said. “Our findings support conducting future human clinical trials to assess the value of estrogen-blocking therapies combined with other therapies to treat bone metastases across multiple cancer types, in both women and men.”
The authors added, “Furthermore, as shown in the final set of experiments, inhibition of ERα in macrophages may not be effective by itself but could synergize with immunotherapies because it facilitates T cell infiltration into static lesions.” The team acknowledged that they didn’t see any synergy between Erαknockout in macrophages and anti-PD1 treatment. However, they noted, “… it is still worth exploring the combinatory effects with other immunotherapies. Therefore, our findings may warrant future clinical trials on combined endocrine and immunotherapies on patients with bone metastases, and this combination may be extended to other cancer types and to patients of both genders.”
The post Method Identifies Cellular Makeup of Microenvironments Favoring Tumor Metastasis appeared first on GEN - Genetic Engineering and Biotechnology News.
Apa Reaksi Anda?
 Suka
0
Suka
0
 Kurang Suka
0
Kurang Suka
0
 Setuju
0
Setuju
0
 Tidak Setuju
0
Tidak Setuju
0
 Bagus
0
Bagus
0
 Berguna
0
Berguna
0
 Hebat
0
Hebat
0